Insulin for People with Type 2 Diabetes
Insulin is a hormone produced by your pancreas. Often referred to as the “most powerful” hormone in the human body, we all need insulin to live. Without enough insulin, your body cannot use the sugar (glucose) in your bloodstream for fuel. Without enough insulin, the amount of sugar in your bloodstream can rise to above-normal levels that threaten your short-term and long-term health.
In people with type 2 diabetes, your body is struggling with two things that can lead to high blood sugar levels:
- Insulin resistance: This is when your body isn’t responding to normal amounts of insulin and isn’t able to use that insulin effectively to manage your blood sugar levels. Some insulin resistance can be improved through lifestyle changes (like physical activity and weight-loss). For some people, insulin resistance is a deeper issue that cannot always be easily changed. This means you may need support from diabetes medications or insulin to help you manage healthy blood sugar levels.
- Beta-cell dysfunction: This is when your body struggles to produce normal amounts of insulin. Beta-cells are produced by the pancreas, then those cells produce insulin. In many people with type 2 diabetes, your body struggles to produce normally functioning beta-cells which means you don’t produce enough insulin. This can worsen over time, and your body is able to produce less and less insulin over time. This means you’ll need more support from diabetes medications or insulin to help you manage healthy blood sugar levels.
After being diagnosed with type 2 diabetes, your doctor will likely prescribe the oral medication metformin first. You’ll also be encouraged to make changes in your food and exercise habits. However, because many people with type 2 diabetes experience gradual beta-cell dysfunction, insulin may eventually be the best medication to help you manage safe and healthy blood sugar levels.
It’s okay if you need insulin!
There is a great deal of shame and guilt that can come with needing insulin as a person with type 2 diabetes. It’s important to note that starting insulin is never the fault of the person with diabetes and that this treatment is to help improve or maintain a high quality of life. Beginning an insulin regimen to manage or improve your blood sugar levels does not mean you have “failed” at diabetes management.
It simply means your body needs insulin to help you manage healthy blood sugar levels.
Today, there are so many types of support and technology to help you live well with diabetes, including long-acting and rapid-acting insulin, inhaled insulin, continuous glucose monitors that check your blood sugar without daily finger pricks, smart insulin pens and insulin pumps!
Check out the resources below to learn more about insulin and how it’s used to manage type 2 diabetes.
Insulin Resources for Type 2 Diabetes Management:


GoodRx Announces New Way for People to Access Lantus for $35
GoodRx partnered with Sanofi to offer $35 insulin coupons to all, regardless of their insurance status, for use at pharmacies nationwide. MORE

Omnipod GO Receives FDA Approval for Adults with Type 2 Diabetes
This FDA approval marks the first-of-its-kind of a basal-only tubeless insulin pod for adults with type 2 diabetes. MORE

This New App is Designed to Make Tracking Your Insulin Easier
If you use Lilly brand insulin, this new connected platform is designed to make tracking your diabetes data a lot easier. MORE

How to Time Your Insulin Dose with Meal Digestion
Learning how to match the timing of your mealtime insulin dose with the digestion of what you’re eating is a key part of managing diabetes.MORE

Switching to an Insulin Patch After 50 Years of Injections
A 66-year-old woman who's been living with diabetes for over 50 years shares her experience using the CeQur Simplicity insulin patch.MORE

What We Can Learn From Diabetes Care in India
Leaders in India discuss the progress they've seen across every facet of diabetes care in the country.MORE

Using Insulin for Type 2 Diabetes: Benefits Beyond Your A1c
Using insulin to manage type 2 diabetes can positively impact more than just your A1c and blood sugar levels. MORE
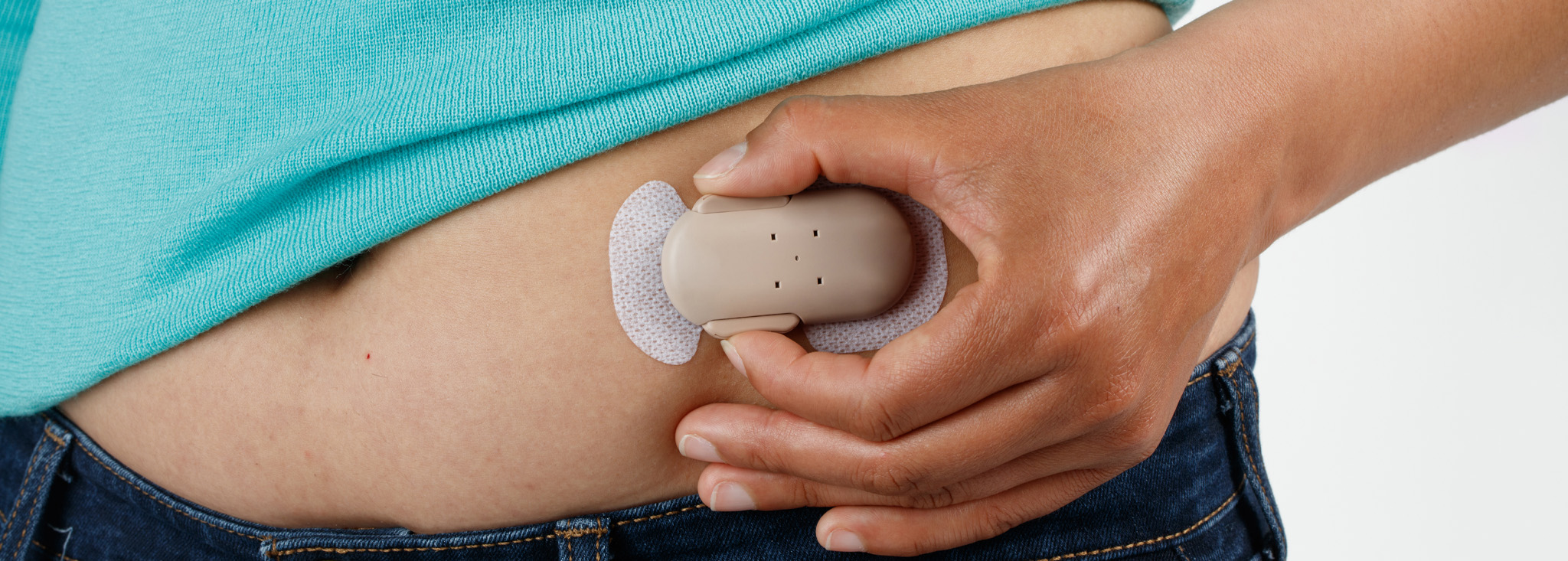
Guide: Asking Your Doctor About Using an Insulin Patch
The CeQur Simplicity insulin patch is so new that your doctor may not even know about it! Learn how to get the conversation started…MORE

All About the CeQur Simplicity Insulin Patch
This ultra-thin wearable insulin patch is a simple and convenient way to deliver your mealtime and correction dose insulin!MORE

If You Take Insulin for Type 2 Diabetes, Your Doctor Should Also Prescribe This…
If you take insulin as a person with type 2 diabetes, your healthcare provider should prescribe emergency glucagon, too. Here’s why…MORE

Insulin Pens vs. Insulin Pumps for Type 2 Diabetes
Using insulin to manage type 2 diabetes comes with options! Here are the pros and cons of insulin pens vs. pumps.MORE

Guide: Talking to Your Doctor About Insulin for Type 2 Diabetes
Starting insulin therapy for type 2 diabetes can be overwhelming—there’s a lot to learn! Here are some questions to ask your doctor to help you get started with using insulin.MORE
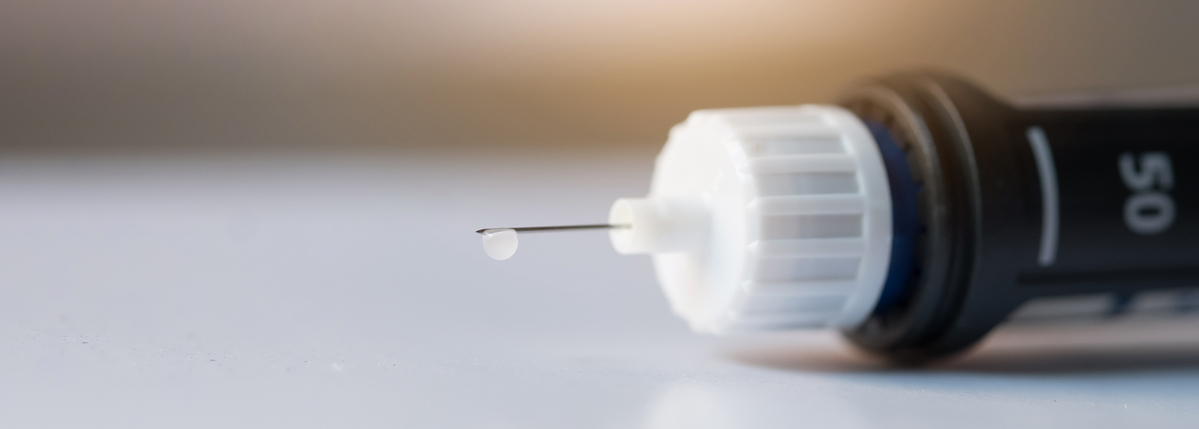
Guide: Using a Smart Insulin Pen to Manage Type 2 Diabetes
Insulin pens can be an incredibly convenient option for people with type 2 diabetes who manage their condition with insulin.MORE
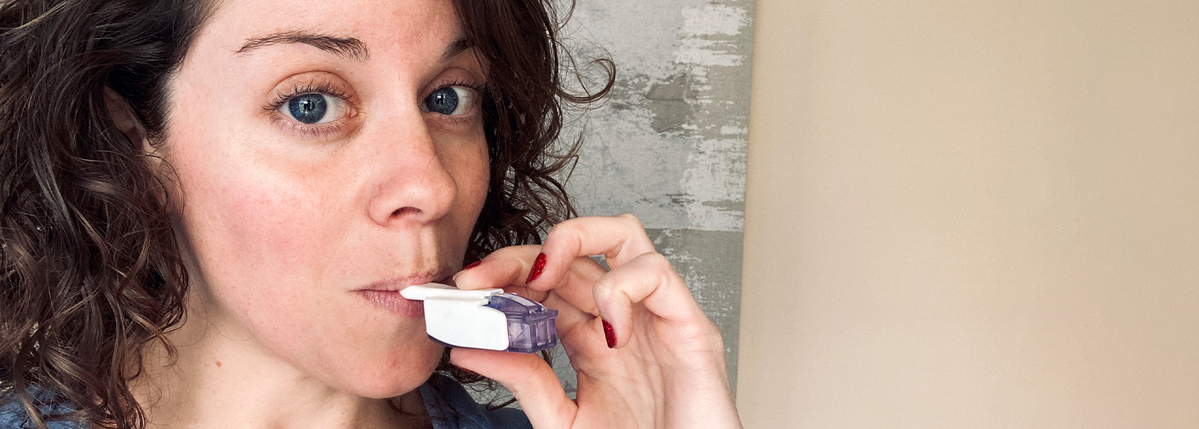
Inhaled Insulin: Myths Vs. Facts
Inhaled insulin has been around for several years, but there are many myths about this zero-poke method of taking insulin.MORE

Talking to Your Doctor About Inhaled Insulin
Your doctor might not know Afrezza's inhaled insulin can be used in Type 1 and Type 2 diabetes. Here are a few tips to start the conversation.MORE

The Benefits of Logging Insulin
Logging your insulin may seem tedious, but it actually has benefits that can improve your Type 2 diabetes management. Read our easy tips here. MORE

How Smartpen Devices Help People with Type 2 Diabetes
Insulin pumps aren't the only option for people with T2D to improve their care. Smartpens are a new way to learn how well your insulin regimen is working.MORE

Logging Insulin Dosages 101
Just starting insulin? This easy guide will help you learn how to log your insulin dosages and when to determine the best times. MORE

Syringes vs. Pens vs. Smart Pens: Pros and Cons
Need a quick breakdown of the pros + cons of syringes, insulin pens, and a smart pen? Learn which insulin delivery method may work for you.MORE

Insulin + Temperature: Why It’s Really Important
Insulin is very sensitive to hot and cold temperatures. Even an hour in a hot car can spoil your insulin. Learn how to keep it safe...MORE

How to Calculate Insulin Dosing for Meals
Taking insulin before meals? Learn how to calculate how much you need before eating using these easy tips. MORE

All About Inhaled Insulin
Here’s everything you need to know about using inhaled insulin as part of your diabetes management routine.MORE

The Type 2 Diabetes Guide to Basal Insulin
Basal insulin works like a base regardless of the food that is consumed and helps regulate blood sugars. Learn more in our guide to manage T2D.MORE

Take Insulin? Four Things You Need to Know Before Your Next Low Blood Sugar
For everyone who doses insulin, low blood sugar (also called hypoglycemia) is the most immediate and severe possible side effect. MORE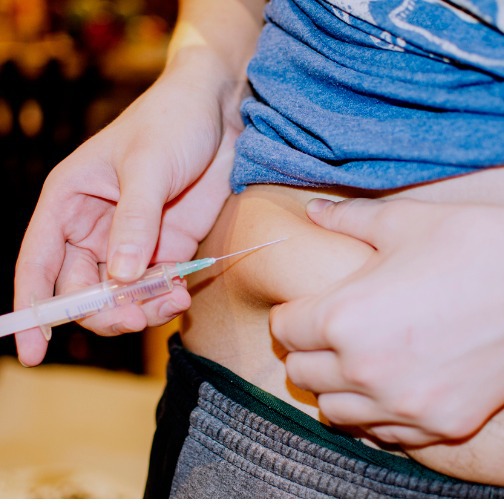
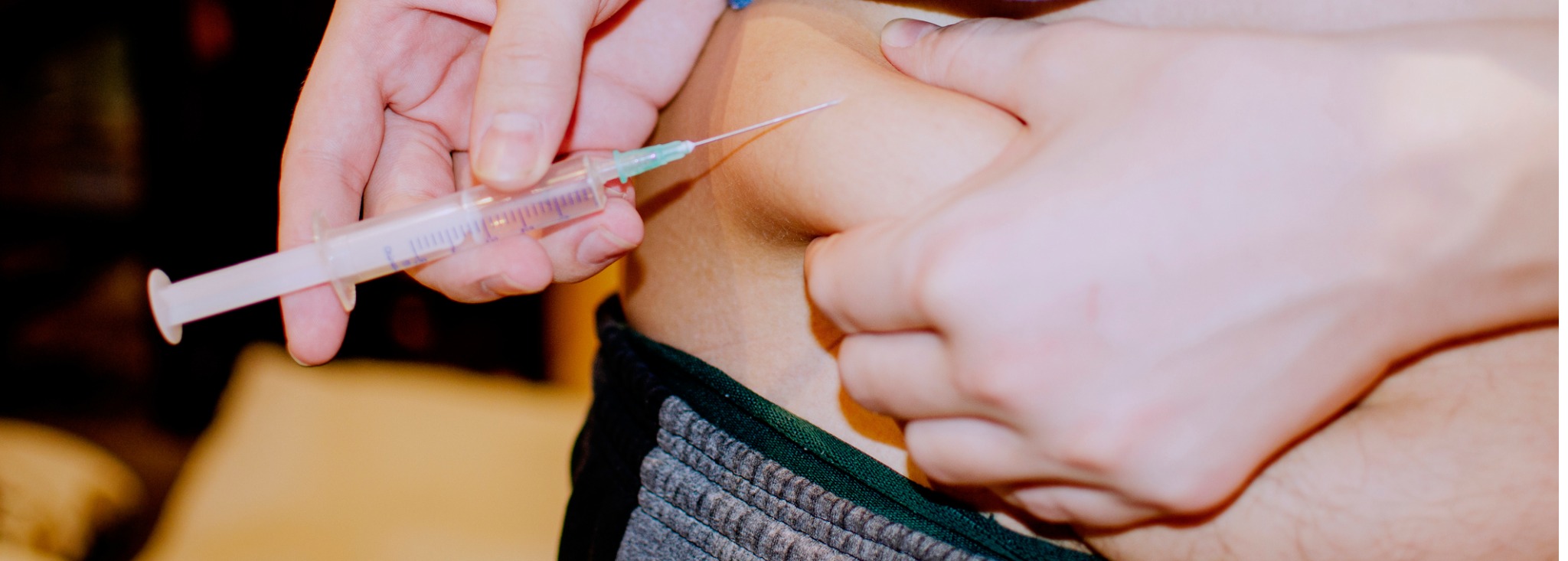
The Type 2 Diabetes Guide to Bolus Insulin
Bolus insulin can help prevent glucose spikes after meals or correct for high blood sugar. Learn more in our guide to manage T2D.MORE
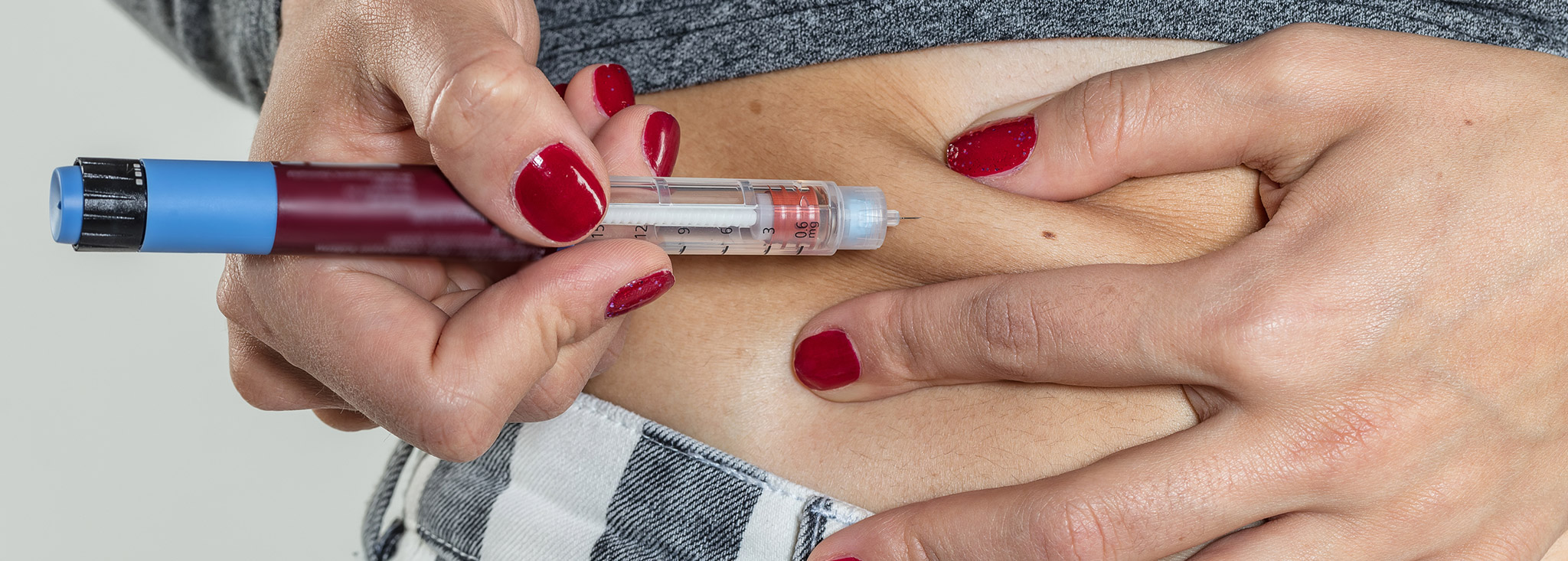
Ask a CDCES: Why Should I Change My Insulin Injection Sites?
Why should you change your insulin injection sites? Get your answer in our Ask a Diabetes Care and Education Specialist (DCES) column!MORE
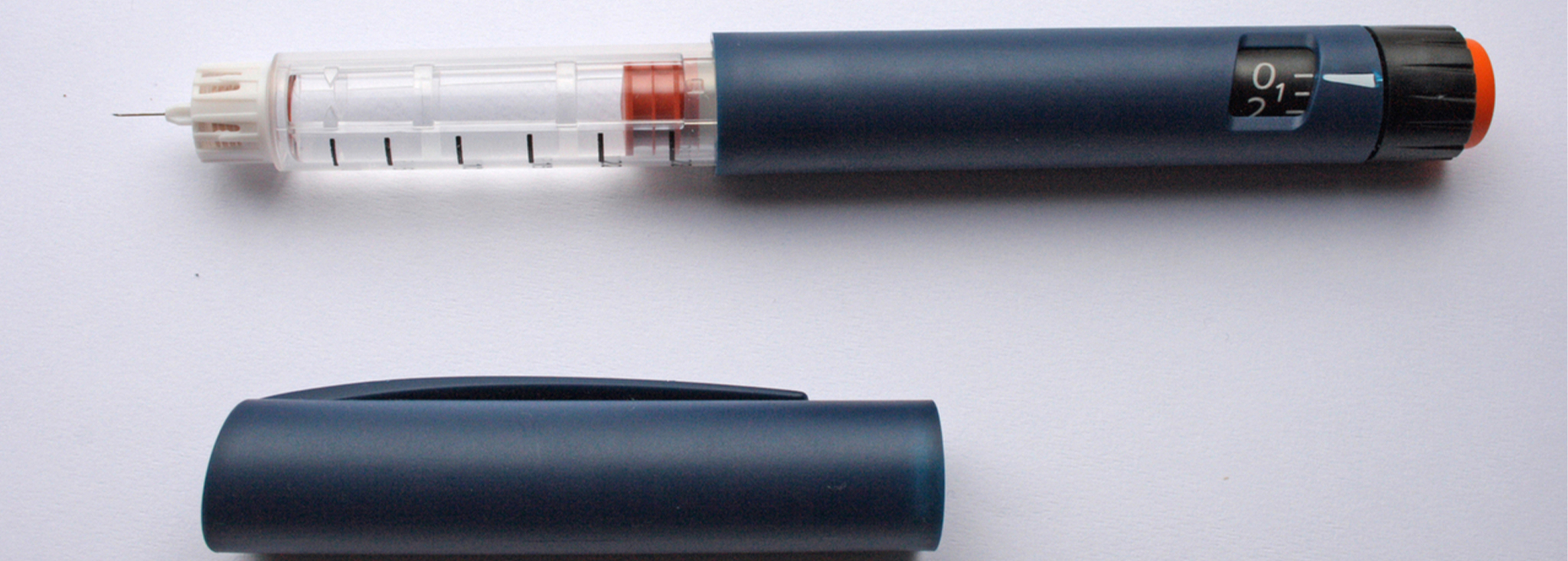
How Insulin Works
Insulin is a hormone that helps regulate your glucose levels. But it has other functions to help maintain your health. Read how insulin works. MORE
Do I Need Insulin?
Did you know that the amount of insulin produced by your pancreas is limited? When should you begin using insulin as part of your treatment? Find some good advice and orientation here. MORE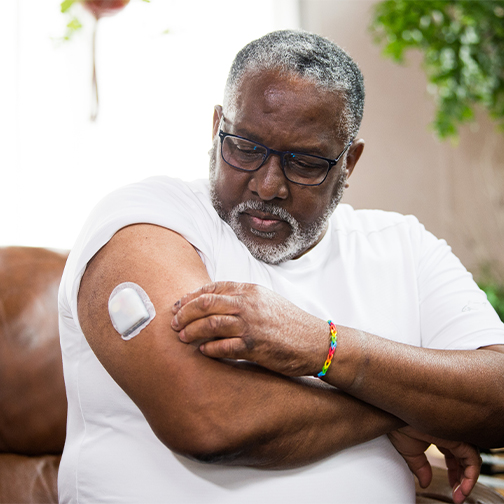
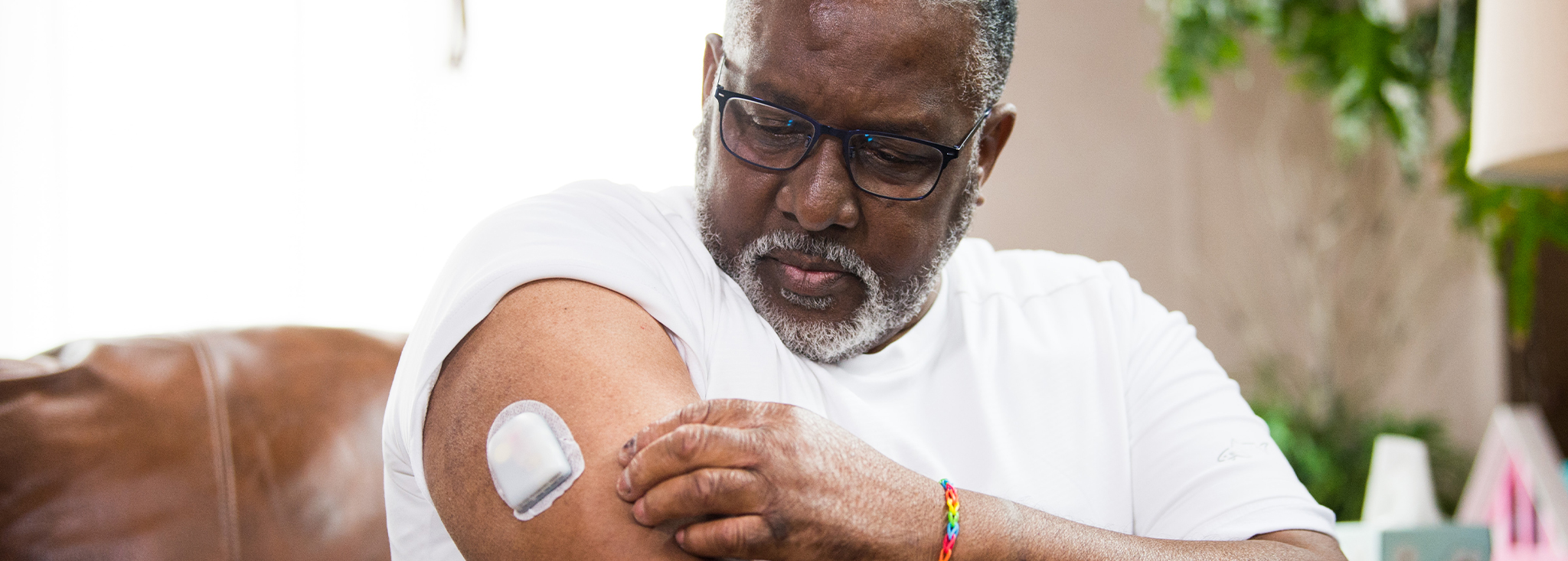
Insulin Pumps for Type 2 Diabetes
Did you know people with T2D can use insulin pumps? Insulin pumps can ease the burden of diabetes self-care. Read about how they can benefit you here.MORE

Making the Switch: From Oral Medications to Insulin
People with type 2 diabetes have a choice in the treatment of their condition, and truly understanding available treatment options is extremely pertinent.MORE
MDI vs Insulin Pumps in Type 2 Diabetes
People with type 2 diabetes have a choice in the treatment of their condition, and understanding any and all options is a vital first step in proper diabetes management.MORE
The Type 2 Guide to Insulin
Understanding the body's relationship with insulin is a crucial part of diabetes education.MORE

How to Give an Insulin Injection
Read our step-by-step guide on how to give an insulin injection to yourself or a person who lives with Type 2 diabetes.MORE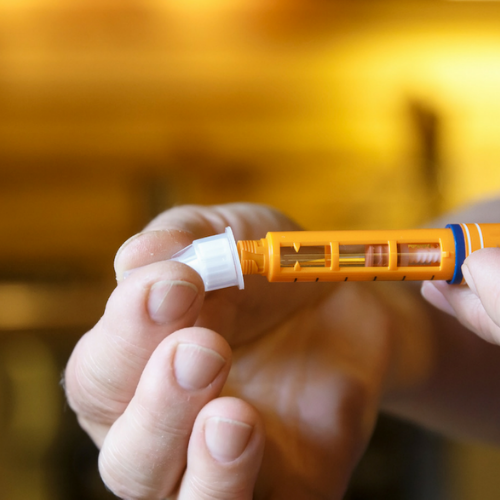
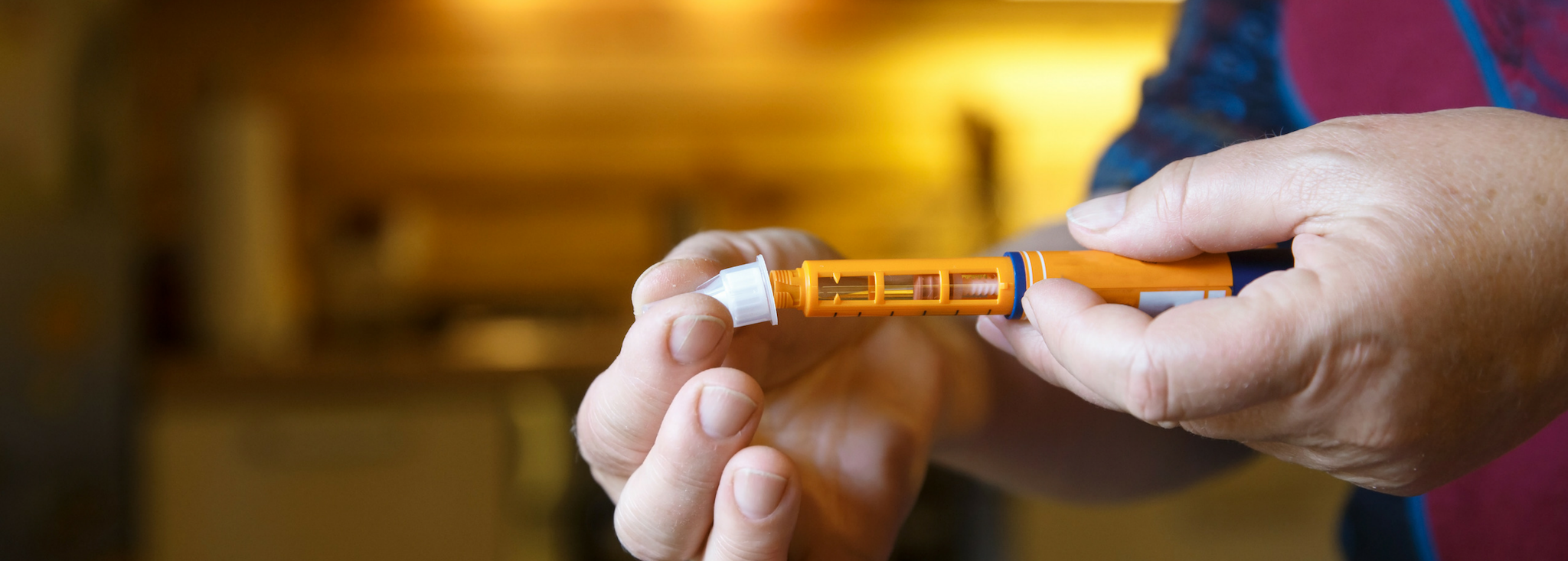
Insulin Delivery Methods
Today, there are a variety of insulin delivery methods to choose from as a person with Type 2 diabetes. Learn about your options here!MORE
Additional Resources and Patient Stories


Insulin Improved My Quality of Life
Alex Charnin shares how she switched doctors to be put on insulin after her endo focused too much on her weight and not her glucose levels.MORE

Former Chef with Type 2 Diabetes Proves Great Food Can Be Healthy + Tasty
Former Chef, Carlos Gonzalez, learned how to make healthy food choices without sacrificing flavor after he was diagnosed with T2D. MORE

I Accidentally Took 18 Units of the Wrong Insulin
Jean Flunker shares how she accidentally took the wrong insulin and her thoughts on insulin stigma in Type 2 diabetes. MORE

A Game Plan to Remember to Take Insulin
Vickie Nell shares her story, the impact of missing insulin dosages, and how she remembers to take insulin. MORE

After Years of Denial, This Man Manages His Diabetes with Insulin
When Arvind Ramaswamy was diagnosed with MODY 1 diabetes at 25 years old in 1995, he was convinced the doctors had made a mistake.MORE

“My doctor refused to put me on insulin for Type 2 diabetes, so I got a new doctor.”
Before Kate Cornell was diagnosed with Type 2 diabetes in 2005 at 50 years old, she had no idea anything was wrong. Today, she uses both metformin and insulin to manage her blood sugars.MORE

“I stopped taking insulin in January 2021,” says Prince Blue
With an A1c of 17.6 percent at his diagnosis, Prince looks back and realizes he should’ve started insulin right away. But today, he’s no longer taking insulin and thriving.MORE

From Syringes to Insulin Pens: Making Type 2 Diabetes Life Easier
Katie Saucedo shares her T2D story, switching from syringes to pens, and discusses T2D stigma and insulin. MORE

A New Way to Deliver the Insulin I Need
Luis Doriocourt shares his experiences switching insulin delivery methods - from syringes and vials to insulin pens to manage T2D. MORE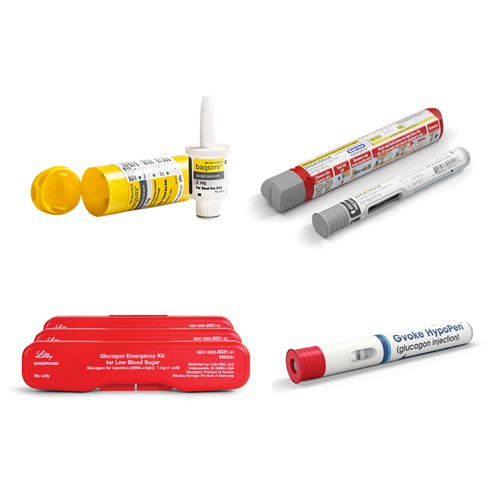
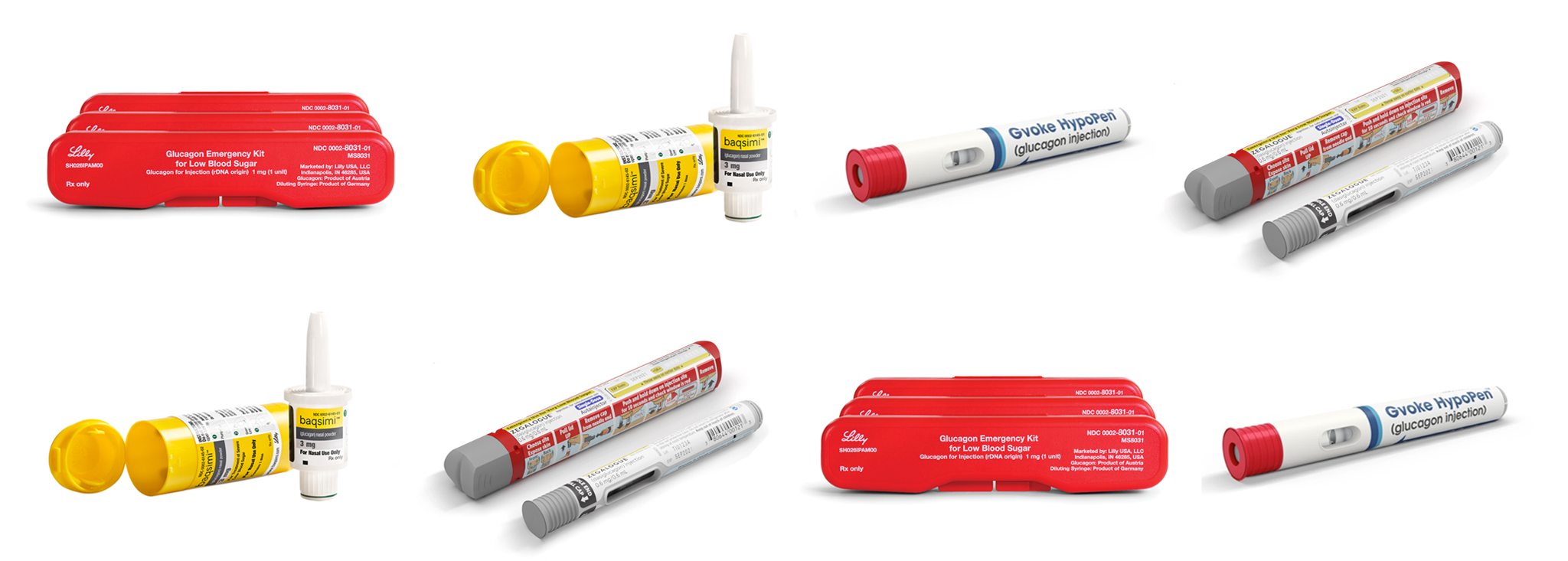
Emergency Glucagon: Everything You Need to Know
Glucagon is a life-saving hormone that anyone with diabetes taking insulin should have on-hand!MORE

Being My Own T2D Success Story with Omnipod
Prince Blue joins Beyond Type 2 to discuss updates to his journey with type 2 diabetes, including finding weight loss success with Omnipod. MORE

Insulin Stigma in the Type 2 Diabetes Community
Insulin stigma in the T2D community is real and negatively impacts diabetes self-care and management. Read how it hurts people with T2D. MORE

Pump Access and Type 2 Diabetes
A person with Type 2 diabetes may eventually require insulin to manage glucose levels. Wearing an insulin pump can make that transition easier and simplify living with diabetes. Learn about pump acces...MORE

Trusting Myself with Insulin
Former pro wrestler, Dan Denton, shares when he first started taking insulin and how he's learned to trust himself with it to manage T2D.MORE

No Shame in Using Insulin
Insulin stigma in T2D care is real and causes people with type 2 to feel like they're failing at managing it. Joseph Martin shares why that doesn't have to be the case. MORE

What to Know About the 2021 Standards of Care for Type 2 Diabetes
It's important to know which guidelines doctors follow to develop your treatment plans. Here's what to know about the 2021 Standards of Care.MORE
When Medications are Prescribed for Type 2 Diabetes Management
Doctors follow a set of protocols to determine when to recommend medications to T2D patients. Learn when they're likely to prescribe them.MORE

10 Misconceptions about Type 2 Diabetes
There are many things people get wrong about type 2 diabetes. Here are 10 misconceptions about diabetes. MORE

The Impact of Insulin Rationing on Mental Health
Insulin rationing is an unfortunate reality for people with diabetes and the burden on mental health is something people with diabetes shouldn't have to bear. MORE

Hypoglycemia Awareness and Type 2 Diabetes
High blood sugar isn't the only thing people with diabetes need to watch out for, but low blood sugar, too. Boost your awareness about hypoglycemia here!MORE

Research Shows Fridges Might Be Destroying Insulin
A 16-month study has shown that 79% of insulin is kept at temperatures outside of the recommended range. Insulin stored in the fridge is at a much higher risk. MORE
Discounts on Insulin
These prescription assistance programs can help make your insulin prescription more affordable.MOREEducational content related to insulin use and insulin logging methods for Type 2 diabetes is made possible with support from Lilly Diabetes.




