Study Shows Combination Therapy Leads to Better Outcomes for Newly Diagnosed T2D Patients
Written by: Todd Boudreaux
3 minute read
September 18, 2019
The new study shows combination therapy in patients newly diagnosed with type 2 diabetes provides better outcomes than metformin alone.
On September 18, Novartis announced results from the Phase IV VERIFY study (Vildagliptin Efficacy in combination with metfoRmIn For earlY treatment of type 2 diabetes). Results showed dual-combination therapy using vildagliptin and metformin at diagnosis of type 2 diabetes (T2D) led to better outcomes than treating with metformin alone.
The findings were presented at the EASD 2019 annual conference in Barcelona, Spain and published in The Lancet. For decades metformin has been the first-line treatment for T2D and other medications are typically prescribed only when monotherapy with metformin fails to adequately treat the disease. This study is the first of its kind to show that combination therapy in newly-diagnosed patients should be considered the standard of care.
“The initial findings from the VERIFY study uniquely demonstrate that early intervention with a combination therapy strategy provides greater and durable long-term benefits for patients. The currently recommended initial monotherapy approach with later treatment intensification in type 2 diabetes management is now shown to be an inferior strategy,” said Professor David Matthews, EASD president and emeritus professor of Diabetic Medicine, University of Oxford, UK.
Vildagliptin (brand names Galvus and Zomelis) is an oral drug used to treat type 2, and belongs to the class of drugs known as dipeptidyl peptidase-4 (DPP-4) inhibitors. DPP-4 inhibitors work by promoting insulin secretion and decreasing the production of glucagon.
See Beyond Type 2’s Guide to Oral Medications Here
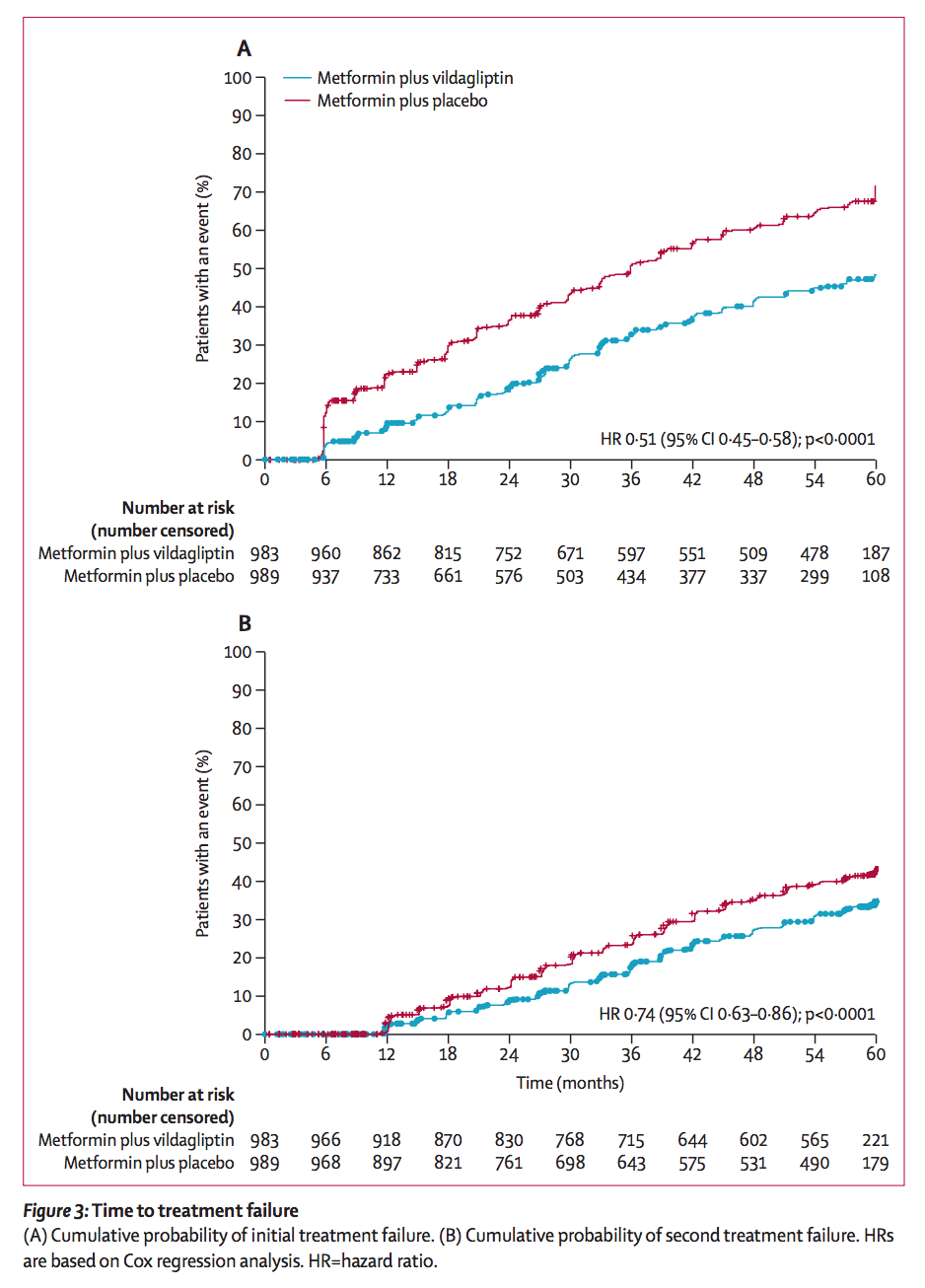 The study was conducted over a five-year period and involved 2001 participants recently diagnosed with T2D, with enrollment beginning in March 2012 and finishing in April 2014. Patients received either combination treatment with metformin and vildagliptin 50 mg twice daily, or standard-of-care initial metformin monotherapy and placebo twice daily.
The study was conducted over a five-year period and involved 2001 participants recently diagnosed with T2D, with enrollment beginning in March 2012 and finishing in April 2014. Patients received either combination treatment with metformin and vildagliptin 50 mg twice daily, or standard-of-care initial metformin monotherapy and placebo twice daily.
If patients did not maintain an HbA1c below 7.0 percent in two consecutive visits at 13-week intervals, this was considered treatment failure. Patients taking metformin and a placebo then received vildagliptin 50 mg twice daily in place of the placebo, while patients in the combination group continued on with their combination therapy. Those receiving the combination therapy from the start were 50 percent less likely to lose blood sugar control than those on metformin alone.
During the second phase of the trial when patients in both groups could be receiving combination therapy, the risk of losing blood sugar control (going above HbA1c 7.0 percent twice) was reduced by 26 percent among those who began on combination therapy, compared to those who transferred after their metformin monotherapy treatment failed. This last statistic is perhaps the most significant, demonstrating that starting on combination therapy can have lasting effects and better outcomes than shifting to combination therapy only after loss of blood sugar control (as defined by the study).
“Despite type 2 diabetes having become an epidemic with growing mortality and morbidity rates, there is a distinct lack of optimized management strategies at diagnosis that can induce durability and slow down disease progression,” said Marcia Kayath, global head medical affairs and chief medical officer, Novartis Pharmaceuticals. “These promising results from the VERIFY study have the potential to improve patient outcomes and the way in which we treat type 2 diabetes in the future”.
Additional analysis of the VERIFY study is ongoing, and more data will be disclosed in journals and at conferences in the coming months.
This article is part of our EASD 2019 coverage. Click here to see all the breaking news from the international diabetes conference.

Author
Todd Boudreaux
<a href="https://beyondtype1.org//leadership/todd-boudreaux/">Todd</a> was diagnosed with type 1 diabetes in 2000, and has been unofficially advocating for type 1 diabetes (T1D) ever since. Before joining the team at Beyond Type 1, Todd wrote and produced television shows for Discovery Channel, Travel Channel and Animal Planet. When he’s not in the office, you can usually find him at a baseball game, traveling or drawing on his Etch A Sketch. You can also <a href="https://www.instagram.com/toddboo/">follow him on Instagram.</a>
Related Resources

Already compatible with Dexcom’s G6 and G7 continuous glucose monitors (CGMs), the Omnipod 5 Automated...
Read more

The younger a person is diagnosed with type 2 diabetes, especially those with obesity, the...
Read more

The Oura Ring, which tracks things like sleep, heart rate, and activity, is joining forces...
Read more

